Nylon represents a versatile synthetic material renowned for its resilience and adaptability in various industrial sectors. Scientists classify nylon as a polymer material characterized by long-chain molecules linked through amide bonds. Synthesis of the substance involves chemical reactions between specific organic monomers derived from industrial feedstocks. Engineering applications frequently utilize Nylon 6 and Nylon 6,6 due to their thermal stability and mechanical strength. Manufacturing processes like injection molding and extrusion convert raw pellets into functional components for diverse industries. Melt viscosity control is important for maintaining quality during polymer processing, such as injection molding and extrusion. Comparisons with materials like acetal and stainless steel highlight the unique weight and resistance profiles of polyamides. Selection criteria focus on moisture absorption and heat tolerance when choosing between different thermoplastic types. Every section explores the manufacturing, composition, and practical benefits of using nylon in modern engineering. Polyamides offer a unique combination of toughness and durability for everyday and industrial products.
What is Nylon?
Nylon refers to a synthetic thermoplastic polymer that belongs to the extensive polyamide (PA) family of materials. Engineering firms value the substance for its exceptional strength and long-lasting durability in demanding environments. Chemical structures within the material provide significant chemical resistance against oils and various industrial solvents. Physical properties include a high degree of toughness, which allows components to withstand repeated impact without failure. Low coefficients of friction create excellent wear resistance for moving mechanical parts like gears and bushings. Modern manufacturing relies on its characteristics to produce reliable textiles and heavy-duty industrial components. Diverse grades of the material accommodate specific temperature and stress requirements in automotive and aerospace designs. Selection of the correct polyamide ensures optimal performance for specialized mechanical assemblies. Polyamides remain essential for creating resilient fibers used in high-performance outdoor gear and safety equipment.
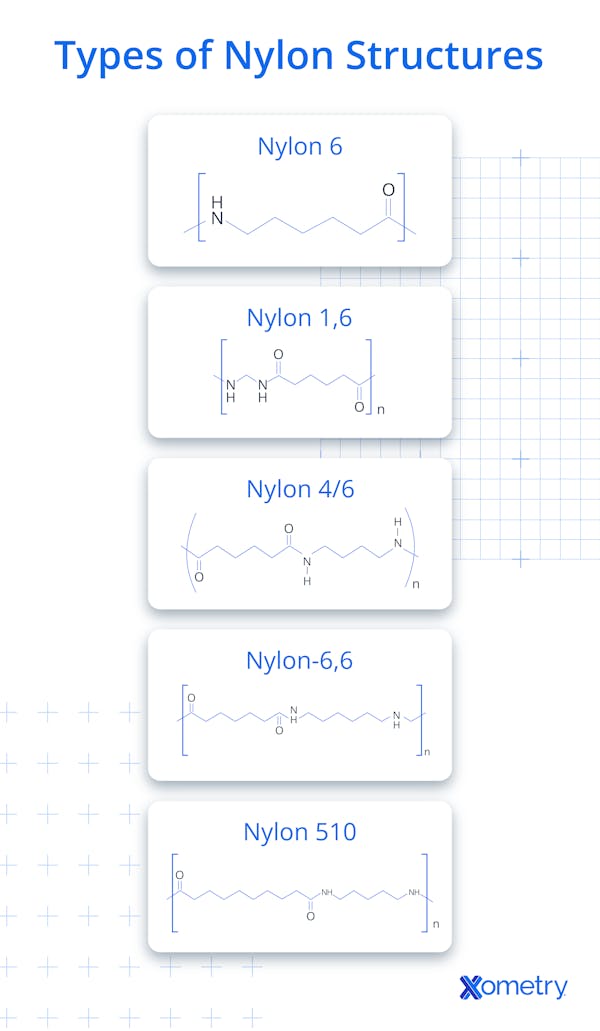
How is nylon classified as a Polymer Material?
Nylon classification as a polymer material depends on its molecular structure, consisting of repeating units joined by covalent chemical bonds. Scientists define polymers as large molecules made from smaller monomeric building blocks through a process called polymerization. Polyamides fit into the thermoplastic category because they soften when heated and solidify upon cooling. Chains of nitrogen and carbon atoms form the backbone of the resilient synthetic substances. Molecular weight distribution influences the physical behavior and mechanical properties of the finished plastic. Linking monomers through amide groups distinguishes polyamides from other polymer families like polyesters or polyolefins. Semicrystalline structures provide the material with its characteristic stiffness and impact resistance. Nylon types are often classified by the number of carbon atoms in their monomers (Nylon 6, Nylon 66), though classification also depends on chemical structure. Industrial classifications separate the polymers based on the number of carbon atoms in their monomer chains. Precise molecular engineering allows for the creation of customized plastic grades for specific industrial needs.
How does Nylon Relate to Polyamide (PA) in Material Classification?
Nylon is a type of polyamide (PA), classified as a synthetic thermoplastic polymer widely used in industry. Chemical compositions involve repeating amide linkages that provide the structural backbone for the entire material group. High strength levels characterize the polyamides, making the chemical compositions ideal for load-bearing structural applications. Manufacturers favor the substance for its durability in harsh outdoor and industrial settings. Resistance to corrosive substances ensures the material provides reliable chemical resistance in automotive fuel systems. High-performance grades offer superior toughness to prevent cracking or deformation under sudden loads. Polyamides demonstrate remarkable wear resistance when subjected to continuous sliding contact. Inherent lubrication properties result in low friction surfaces for high-efficiency mechanical systems. Material science classifies different types based on the specific arrangement of carbon atoms within the repeating units. Broad adoption of the thermoplastics stems from their balanced mechanical and thermal properties.
Is Nylon the Most Widely Used Type of Polyamide?
Yes, nylon is the most widely used type of polyamide in commercial and industrial sectors. Global markets consume vast quantities of the material for textiles, automotive parts, and electrical components. Manufacturers utilize PA6 to achieve excellent surface finishes and reliable processability during high-volume production. Engineering teams specify PA66 for assemblies requiring higher melting points and increased mechanical stiffness. Versatility across different production methods makes the polyamides competitive with many alternative engineering plastics. Cost efficiency and performance balance drive the dominance of the specific polymers in the global supply chain. Consumer products frequently feature the material for its resilience and aesthetic appeal. Widespread availability of raw monomers supports the massive scale of polyamide manufacturing worldwide.
What is Nylon Made Of?
Nylon is made of synthetic polyamide polymers created through chemical reactions between organic monomers derived from petroleum feedstocks. Nylon production involves the linkage of repeating molecular units through amide bonds to form a long-chain polymer structure. Monomers undergo condensation or ring-opening reactions to create the resilient synthetic fibers and plastics. Chemical structures rely on the precise arrangement of nitrogen, carbon, oxygen, and hydrogen atoms. Raw ingredients derive primarily from industrial feedstocks processed through complex refining steps. Specific formulations depend on the intended grade, such as PA6 or PA66 for engineering use. Structural integrity comes from the strong intermolecular forces between the polymer chains. Variations in the chain length affect the mechanical performance and melting temperature of the substance. Industrial synthesis ensures high purity and consistency for large-scale material distribution. Advanced chemistry allows for the inclusion of additives to enhance specific physical properties. Every batch of the material follows strict chemical recipes to maintain its characteristic toughness.
What is the Chemical Composition of Nylon?
Nylon chemical composition involves long-chain synthetic molecules consisting of repeating units linked by strong amide bonds. Nylon can be produced by condensation polymerization (diamines + dicarboxylic acids, e.g., PA66) or ring-opening polymerization (e.g., PA6 from caprolactam). Specific numbers of carbon atoms in the monomers determine the final nomenclature of each nylon grade. Amide groups provide the polar nature that influences moisture absorption and mechanical stiffness. Chemical structures remain stable under moderate heat and exposure to many organic solvents. Nitrogen atoms within the polymer backbone play a crucial role in the formation of hydrogen bonds between chains. Interchain forces contribute to the high melting point and crystallinity of the material. Molecular arrangements vary between different grades to achieve specific flexibility or rigidity levels. Homopolymers and copolymers offer different chemical profiles for specialized engineering requirements. Understanding the atomic makeup helps chemists improve the heat resistance and UV stability of the final product.
Nylon is a widely used engineering polymer valued for its combination of strength, durability, and adaptability across multiple industries. Its semi-crystalline polyamide structure provides high toughness, wear resistance, and low friction, making it suitable for mechanical components such as gears and bearings. The material is lightweight and offers good chemical resistance, which supports its use in automotive, electrical, and industrial applications. However, its performance is significantly affected by moisture absorption, which can reduce stiffness and alter dimensional stability. Nylon also has thermal limitations, as mechanical properties degrade at elevated temperatures compared to metals or high-performance polymers. Despite these constraints, nylon remains a practical material choice due to its balance of performance, cost-efficiency, and ease of manufacturing.
What Raw Materials are Used to Produce Nylon Polymer?
Raw materials used to produce nylon polymer include organic chemical compounds like caprolactam for the synthesis of Nylon 6. Production of Nylon 6,6 requires a combination of adipic acid and hexamethylenediamine through a condensation reaction. The monomers originate from industrial feedstocks derived from large-scale refining operations. Chemical reactions involve the formation of strong amide linkages that create the durable polymer chain. Precise ratios of the ingredients determine the quality and molecular weight of the final plastic. Industrial plants synthesize the precursors using advanced catalytic processes for maximum efficiency. Purity levels of the raw monomers directly impact the transparency and strength of the resulting material. Supply chains for the chemicals remain vital for the global manufacturing of synthetic textiles and plastics. Modern facilities focus on optimizing energy use during the extraction and synthesis of the essential compounds. Scientific advancements continue to refine the methods used to convert raw chemicals into high-performance polymers.
Is Nylon Made From Petrochemical Based Monomers?
Yes, traditional nylon originates from petrochemical-based monomers derived from crude oil refining processes. Industrial synthesis utilizes precursors like benzene and cyclohexane to create the necessary building blocks for polyamides. Global production remains dependent on fossil fuel resources for high-volume manufacturing of standard grades. Petroleum feedstocks provide the consistency and scale required for global supply chains. Bio-based alternatives exist but currently represent a smaller portion of the total market share. Environmental concerns drive research into sustainable sources for the essential chemical monomers. High energy requirements characterize the conversion of crude oil into refined polymer precursors. Manufacturers continue to balance performance needs with the availability of traditional petrochemical ingredients.
How is Nylon Manufactured in Industrial Production?
Nylon is manufactured in industrial production by polymerization reactions that combine specific chemical monomers into long polyamide chains. Nylon manufacturing in industrial production involves complex polymerization reactions like condensation or ring-opening processes. Facilities heat raw monomers until they react to form long-chain molecules with high molecular weight. Molten polymer passes through extrusion dies to create continuous strands of the synthetic substance. Cooling baths solidify the material before it undergoes pelletization for easy transport and storage. The pellets serve as the primary raw material for injection molding and fiber spinning operations. Quality control measures monitor temperature and pressure during every stage of the chemical reaction. Vacuum systems remove moisture and volatile byproducts to ensure the structural integrity of the plastic. Industrial scales allow for the production of thousands of tons of material annually. Advanced automation systems regulate the flow of chemicals to maintain consistent polymer properties throughout the batch. Final products include a wide range of shapes from fine filaments to thick industrial plates. Polyamides provide the necessary properties for creating durable and long-lasting components.
How does Nylon Processing Work in Plastic Injection Molding?
Nylon processing in injection molding begins with heating properly dried pellets to a molten state. High-pressure rams or screws inject the liquid polymer into a precision-machined mold cavity. The material fills every detail of the mold before cooling and solidifying into a functional part. Precise temperature settings prevent thermal degradation while ensuring adequate flow throughout the tool. Moisture management remains critical because wet nylon causes surface defects and reduces mechanical strength. Mold cooling systems regulate the solidification rate to minimize internal stresses and warping. Ejection pins remove the finished component once the plastic reaches the required hardness. Operators adjust cycle times to maximize production efficiency and maintain dimensional accuracy. Specialized equipment must handle elevated processing temperatures (typically ~220–300°C depending on grade). Successful molding results in durable parts for automotive, electrical, and consumer goods sectors.
Is Nylon Suitable for Plastic Injection Molding Applications?
Yes, nylon is suitable for injection molding applications due to its excellent flow characteristics and mechanical performance. Manufacturers utilize the material for producing complex gears, bushings, and automotive engine housings. High strength and wear resistance ensure molded components perform reliably under significant stress. Nylon can achieve good dimensional accuracy, but moisture absorption and shrinkage can reduce long-term dimensional stability. Polyamides adapt well to the high pressures and temperatures found in modern molding machinery. Low-friction properties make the material ideal for moving parts in mechanical assemblies. Widespread use in industrial sectors confirms the versatility of the material for molded products. Optimizing the molding process results in high-quality parts depends on mold design, processing, and crystallinity surface finishes.
Why is Viscosity Important When Processing Nylon?
The importance of viscosity in nylon processing relates to the ability of the molten polymer to flow into complex mold geometries. Proper flow levels ensure that every corner of the mold cavity fills completely without forming air pockets or voids. Inconsistent viscosity leads to defects like short shots or excessive flashing on the part edges. Molecular weight and temperature directly influence how thick or thin the liquid plastic becomes during heating. High-performance molding requires a stable viscosity to maintain dimensional accuracy across large production runs. Operators monitor flow rates to prevent internal stresses that cause warping or structural failure. Low viscosity allows for the production of thin-walled components with great detail. Excessive temperature reduces viscosity and can cause thermal degradation, which reduces mechanical properties. Understanding the fluid dynamics helps manufacturers optimize cycle times and reduce production waste. Precise control over material flow ensures the final product meets all engineering specifications for durability.
How does Viscosity Affect Nylon Melt Flow During Molding?
Viscosity affects nylon melt flow by determining the resistance of the liquid polymer to movement within the injection system. Higher viscosity levels reduce the speed at which the material travels through runners and gates. Lower viscosity improves flowability for intricate parts with thin cross-sections. Melt flow behavior influences the surface finish and internal density of the molded component. Processing temperatures play a major role in thinning the polymer for better mold penetration. Molecular weight distribution dictates the inherent thickness of the melted substance. Faster fill rates occur when the viscosity remains within the optimal range for the specific machinery. Inconsistent flow causes variations in part weight and structural strength between cycles. Monitoring the melt flow index provides data for adjusting production parameters. Achieving the correct balance of flow ensures the production of high-quality industrial parts.
Does Nylons Viscosity Influence Injection Pressure and Fill Rate?
Yes, nylon’s viscosity influences the required injection pressure and the fill rate during manufacturing. High viscosity levels necessitate greater pressure from the machine to move the molten plastic into the tool. Increased injection pressure results in higher internal stresses within the finished part. Lower viscosity allows for faster fill rates and shorter production cycles. Optimizing the relationship between pressure and flow improves the consistency of the final product. Changes in processing temperature modify the viscosity to suit specific mold designs. Sink marks and voids are primarily influenced by packing pressure, cooling, and shrinkage; viscosity plays a secondary role. Manufacturers achieve better efficiency by matching the polymer flow characteristics to the equipment capabilities.
What Are the Types of Nylon Used in Engineering Applications?
The types of nylon used in engineering applications are shown in the table below.
| Type | Common Name | Key Property |
|---|---|---|
Type Nylon 6 | Common Name Polycaprolactam | Key Property High impact strength and surface finish |
Type Nylon 6,6 | Common Name Polyhexamethylene Adipamide | Key Property Nylon 6,6 has a higher melting point and stiffness than Nylon 6, but also improved creep resistance. |
Type Nylon 11 | Common Name Polyamide 11 | Key Property Excellent chemical resistance and bio-based |
Type Nylon 12 | Common Name Polyamide 12 | Key Property Very low moisture absorption and high dimensional stability |
Type Glass Filled | Common Name Reinforced Nylon | Key Property Superior structural rigidity and heat resistance |
The table presents major nylon categories used in engineering materials. Nylon 6 delivers strong impact resistance and maintains a smooth molded surface for manufactured parts. Nylon 6,6 provides a higher melting temperature and stronger stiffness that supports structural mechanical components. Nylon 11 supplies strong chemical resistance and originates from castor oil feedstock used in bio-based polymer production. Nylon 12 provides very low moisture absorption and maintains dimensional stability in precision parts. Glass filled nylon contains glass fiber reinforcement that increases structural rigidity and improves heat resistance in industrial components within Types of Nylon.
1. Nylon 6 (Polycaprolactam)
Nylon 6 offers strong impact resistance and high flexibility compared with many polyamide grades. Surface finishes remain smooth and visually clean for consumer-facing products. Manufacturing teams select Nylon 6 for heavy duty power tool housings and automotive interior components. Injection molding supports fast production cycles in large scale industrial manufacturing. Mechanical assemblies rely on the fatigue resistance of Nylon 6 during repeated stress cycles, known as Nylon 6 (Polycaprolactam).
2. Nylon 6,6 (Polyhexamethylene Adipamide)
Nylon 6,6 features a high melting temperature and strong mechanical stiffness for structural engineering parts. Abrasion resistance supports use in gears and sliding mechanical components under continuous friction. Automotive manufacturers specify Nylon 6,6 for under hood components exposed to engine heat. Low creep behavior preserves part geometry under constant load conditions. Chemical exposure from oils and lubricants produces minimal degradation in Nylon 6,6 (Polyhexamethylene Adipamide).
3. Nylon 11
Nylon 11 provides strong chemical resistance and maintains flexibility under very low temperature conditions. Raw material production relies on castor oil feedstock that supports biobased polymer manufacturing. Fuel lines, hydraulic tubing, and protective industrial coatings rely on Nylon 11 for durability. Nylon 11 has good impact resistance and retains toughness at low temperatures, making it suitable for outdoor applications. Outdoor mechanical systems benefit from the high impact strength of Nylon 11.
4. Nylon 12
Nylon 12 exhibits very low moisture absorption among commercial polyamide materials. Dimensional precision remains stable during temperature shifts and humidity changes. Electrical insulation performance supports use in cable jackets and electronic connectors. Stress cracking resistance extends the life of pressurized fluid systems. Medical instruments and technical devices rely on the consistent toughness of Nylon 12.
5. Glass Filled Nylons
Glass filled nylons contain microscopic glass fibers that strengthen the polymer matrix. Structural rigidity increases compared with unreinforced polyamide plastics. Heat deflection temperature rises and allows operation under higher thermal loads. Shrinkage during cooling decreases due to fiber reinforcement that stabilizes the polymer structure. Automotive intake manifolds and industrial structural brackets rely on reinforced materials known as Glass Filled Nylons.
How Does Nylon Compare to Other Engineering Plastics in Material Selection?
Nylon offers high toughness and wear resistance, though some materials (UHMWPE, PEEK) may exceed it in specific conditions. Material selection processes prioritize polyamides for applications requiring a balance of strength and cost efficiency. Many alternatives lack the same fatigue performance found in high-grade synthetic polyamides. Moisture absorption remains a key differentiator as nylon picks up more water than many other thermoplastics. High melting points allow for use in environments where cheaper plastics would soften. Selection criteria involve evaluating the impact resistance versus the dimensional stability of the part. Polyamides compete effectively with polycarbonates in terms of durability for industrial housings. PEEK offers better high-temperature performance but comes at a higher cost than standard nylons. Versatility in manufacturing makes polyamides a frequent choice for diverse industrial sectors. Engineers favor the materials for their ability to withstand harsh chemicals and repeated friction.
What Are the Differences Between Nylon and Acetal (POM) in Precision Applications?
The differences between nylon and acetal (POM) in precision applications are listed below.
- Acetal (POM): Acetal provides superior dimensional stability due to its extremely low moisture absorption rates. The material offers higher stiffness and a lower coefficient of friction for precision gears. Chemical resistance remains high for fuel and solvent exposure in mechanical systems.
- Impact Strength: Nylon offers better impact strength and toughness for parts subject to sudden shocks. Heavy-duty applications favor polyamides for their ability to absorb energy without fracturing.
- Environmental Stability: Acetal maintains better structural integrity in wet environments compared to standard polyamides. Precision components stay within tight tolerances regardless of humidity levels.
- Cost Efficiency: Nylon proves more cost-effective for large-scale structural components that do not require tight tolerances. High-volume production benefits from the lower price point of standard nylon grades than Acetal (POM).
Is Nylon More Moisture Absorbent Than Acetal?
Yes, nylon absorbs more moisture than acetal during long-term exposure to humid environments. Water molecules penetrate the polymer structure of polyamides and behave like a plasticizer. Increased moisture levels lead to dimensional swelling and a reduction in mechanical stiffness. Acetal remains highly stable and resists changes in size when submerged in water. Engineering designs must account for the changes in nylon to prevent part failure. Choosing acetal ensures better precision for components operating in high-humidity settings. Every designer evaluates environmental conditions before selecting the appropriate thermoplastic. Nylon typically loses stiffness and strength but gains toughness and impact resistance when wet. Reliability in precision assemblies requires an understanding of how moisture impacts material performance.
What Are the Advantages and Limitations of Nylon as a Material?
The advantages and limitations of nylon as a material are listed below.
- High Strength: Polyamides provide exceptional load-bearing capabilities for structural and mechanical components. Heavy-duty designs utilize the material to replace heavier metal parts.
- Wear Resistance: Low friction surfaces ensure long-lasting performance in gears and sliding assemblies. Moving parts operate smoothly without the need for constant external lubrication.
- Chemical Resistance: Resistance to oils and solvents protects parts in automotive and industrial environments. Most organic chemicals do not degrade the structural integrity of the polymer.
- Moisture Absorption: Sensitivity to humidity causes dimensional changes and alters physical properties over time. Parts grow slightly larger when exposed to water or high humidity.
- Thermal Limits: Standard grades have lower heat resistance compared to high-performance polymers like PEEK. Components lose stiffness as they approach their specific melting point.
- UV Sensitivity: Prolonged exposure to sunlight degrades the material without the addition of stabilizers. Outdoor applications require specialized grades to prevent brittleness.
How does Nylon Compare to Stainless Steel (SS) in Weight and Corrosion Resistance?
Nylon comparison to Stainless Steel (SS) highlights the massive weight reduction possible when switching to high-strength polymers. Polyamides are lighter than metal alloys, which improves fuel efficiency in transportation sectors. Nylon does not corrode, but it can degrade under UV exposure, hydrolysis, or certain chemicals. Stainless steel offers far greater heat resistance and structural strength for high-load applications. Engineering choices depend on whether the part requires the rigidity of steel or the lightweight nature of plastic. Nylon provides excellent electrical insulation, while metals conduct electricity and heat. Cost advantage depends on production volume; injection molding has high initial tooling cost but low per-part cost at scale. Nylon and stainless steel (SS) materials offer durability but serve different roles based on thermal and mechanical needs.
Is Nylon Lighter but Less Heat Resistant Than Stainless Steel?
Nylon is much lighter than stainless steel but offers lower heat resistance for industrial applications. Polyamides provide a density far below that of high-strength metal alloys for weight-sensitive designs. Melting points for synthetic polymers remain well below the levels where stainless steel begins to soften. Metals retain structural strength at elevated temperatures far better than thermoplastics, especially above polymer softening temperatures. Engineering teams select nylon for lightweight mechanical components that operate at moderate temperatures. Stainless steel remains the preferred choice for high-load structural frames and extreme thermal environments. Nylon and stainless steel materials contribute to modern engineering through their unique physical and chemical profiles.
Summary
This article provided a summary of the uses, properties, types, and advantages of nylon as a material for use in product and part fabrication.
Xometry offers injection molding services for all of your prototyping and production needs. Visit our website to learn more and to request a free, no-obligation quote using our Instant Quote Engine.
Disclaimer
The content appearing on this webpage is for informational purposes only. Xometry makes no representation or warranty of any kind, be it expressed or implied, as to the accuracy, completeness, or validity of the information. Any performance parameters, geometric tolerances, specific design features, quality and types of materials, or processes should not be inferred to represent what will be delivered by third-party suppliers or manufacturers through Xometry’s network. Buyers seeking quotes for parts are responsible for defining the specific requirements for those parts. Please refer to our terms and conditions for more information.


